Choosing an SCIg Infusion vs IVIg Infusion
SCIg vs IVIg: Differences between routes of administration
| IVIg | SCIg | ||
|---|---|---|---|
| Who |  |
Infusion usually performed by an HCP | Infusion can be performed by patients or caregivers after training |
| Where |  |
Is usually performed in a healthcare facility | Once trained, treament almost anywhere (home, office, travel) |
Studies have demonstrated that SCIg delivery is feasible, safe, efficient and cost-effective1-4
SCIg infusions have steadily replaced IVIg infusions1
Many patients prefer SCIg for multiple reasons:1-4
Home
School
Office/Work
Travel
- Ability to self-administer or administration by the patients’ caregiver after training almost anywhere: home, school, office, work, travel, etc.
- Allows greater freedom and convenience
- Minimal impact on family, work, and school activities
- Removes the need to travel to infusion clinics or hospital
Cutaquig allows patients to comfortably switch from IVIg to SCIg, enabling the transition to home care.5
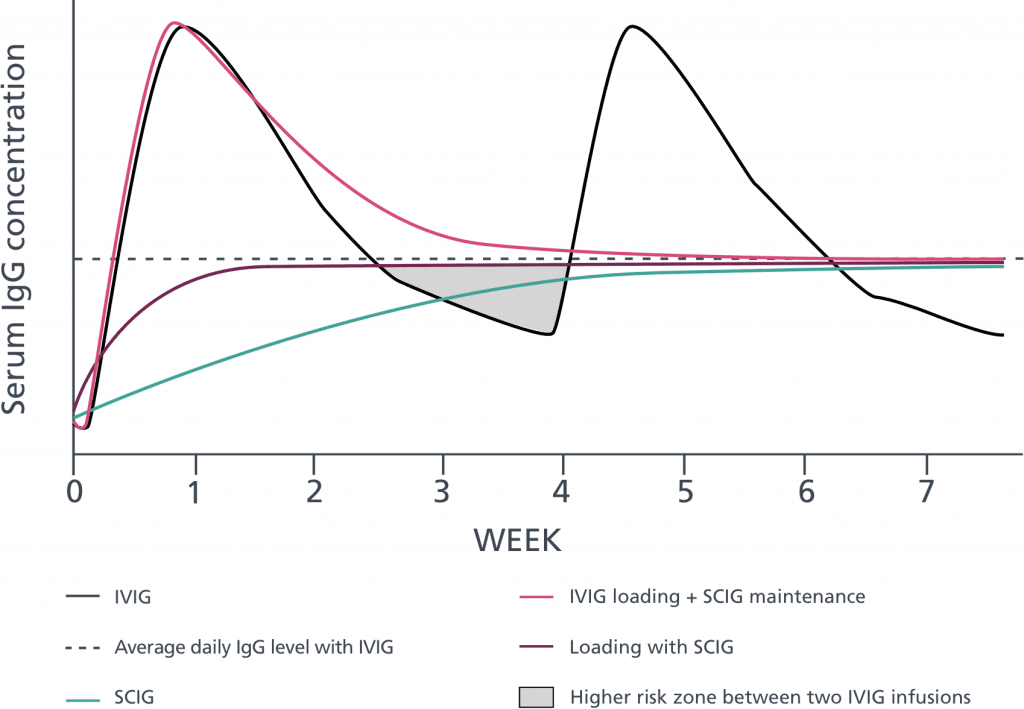
IVIg vs SCIg Infusion: Concentrations over time
SCIg treatment provides stable serum immunoglobulin levels6
- Traditionally, maintenance replacement SCIg therapy is preceded by a switch from existing IVIg therapy
- In most cases, the first SCIg infusion is given 1 week after the last IVIg infusion in order to maintain high serum immunoglobulin levels
- Thereafter, the average daily immunoglobulin level achieved with IVIg can be maintained with regular subcutaneous infusions
References:
- Kobayashi RH, Gupta S, Melamed I, et al. Clinical Efficacy, Safety and Tolerability of a New Subcutaneous Immunoglobulin 16.5% (octanorm [cutaquig®]) in the Treatment of Patients with Primary Immunodeficiencies. Front Immunol. February 2019 | Volume 10 | Article 40.
- McCormack PL. Immune Globulin Subcutaneous (Human) 20% In Primary Immunodeficiency Disorders. Drugs. 2012; 72 (8): 1087-1097.
- Kobrynski L. Subcutaneous immunoglobulin therapy: a new option for patients with primary immunodeficiency diseases. Biologics: Targets and Therapy.2012:6 277–287.
- Berman K. Safety, Efficacy, Tolerability, Advantages and Disadvantages of Intravenous and Subcutaneous Immune Globulin Therapy. Highlights from IG Living Teleconference December 10, 2015. http://www.igliving.com/life-with-ig/teleconference/advantages-and-disadvantages-of-intravenous-and-subcutaneous-immune-globulin-therapy.html.
- Data on file, Octaphama.
- Jolles S, Stein MR, Longhurst HJ et al. New Frontiers in Subcutaneous Immunoglobulin Treatment. Biol Therapy. 2011; 1: 3. https://doi.org/10.1007/s13554-011-0009-3.